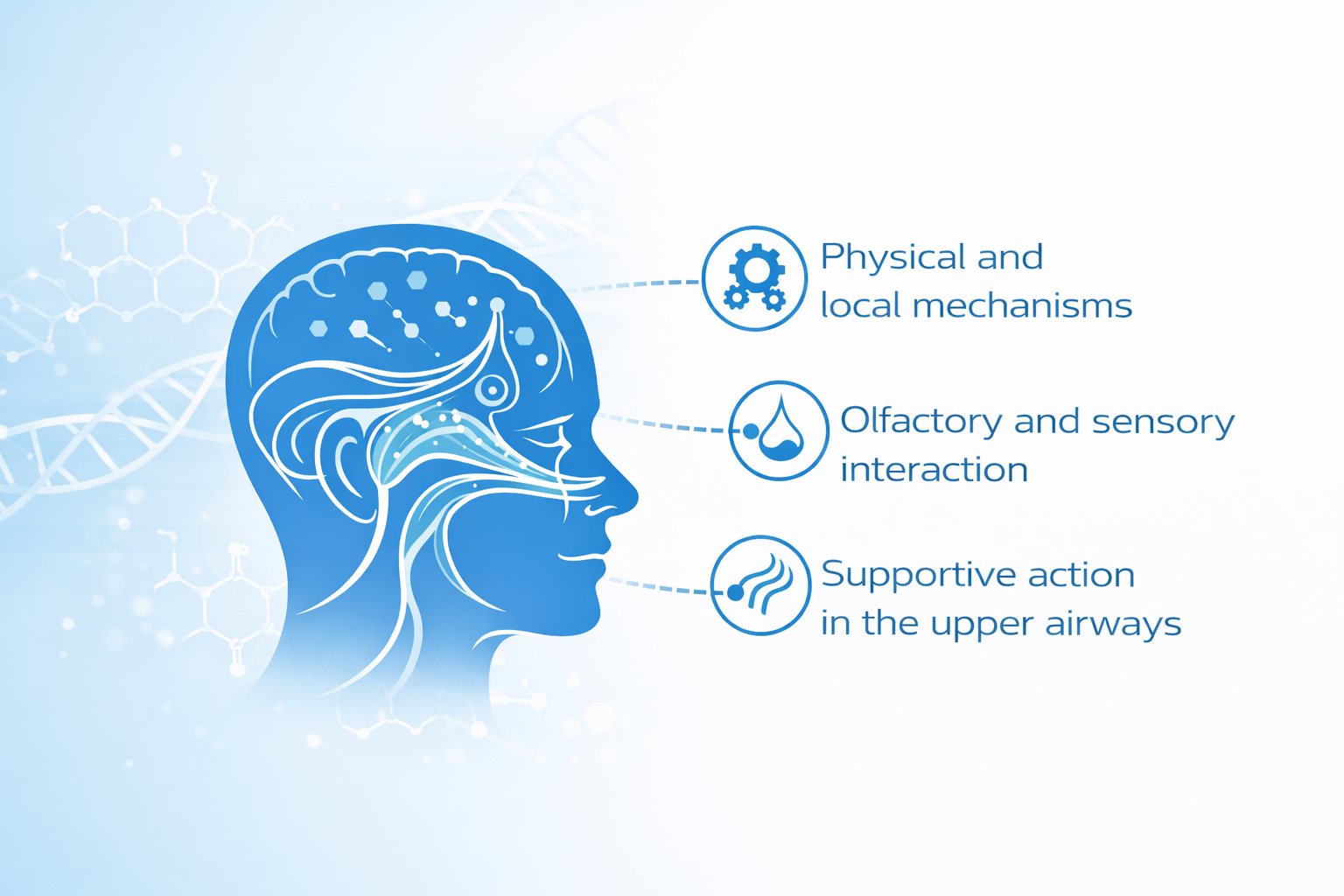
A non-pharmacological support system designed for professional migraine recommendation
SinaVita® Migraine Support is a structured, non-drug product system developed for pharmacies and healthcare professionals seeking compliant, supportive options for customers experiencing migraine and recurring headache.
The system is designed for professional recommendation, either as a complement to standard treatment or as a non-pharmacological first-line support where appropriate.